Potassium nitrate, #"KNO"_3#, is a soluble ionic compound that dissociates completely in aqueous solution to form potassium cations, #"K"^(+)#, and nitrate anions, #"NO"_3^(-)#.
#"KNO"_ (3(aq)) -> "K"_ ((aq))^(+) + "NO"_(3(aq))^(-)#
Now, the solubility of potassium nitrate depends on the temperature of the water, as you can see in the salt's solubility curve
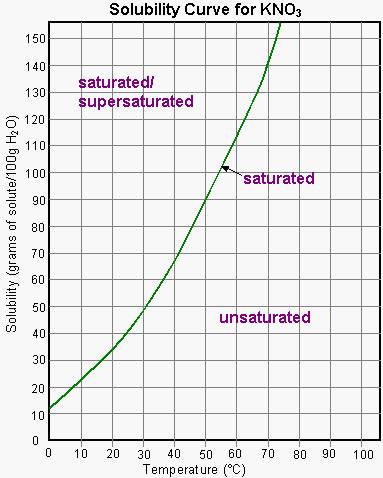
As you can see, potassium nitrate has a solubility of about #"35 g"# per #"100 g"# of water at #20^@"C"#.
Adding this much potassium nitrate per #"100 g"# of water at this temperature will result in the formation of a saturated solution, as you can see by tracing the solubility curve shown in the above image.
In other words, you can only hope to dissolve #"35 g"# of potassium nitrate per #"100 g"# of water at #20^@"C"#. Adding less than #"35 g"# would result in the formation of an unsaturated solution, i.e. a solution that can dissolve more solute.
Likewise, adding more than #"35 g"# would keep the solution saturated. The excess potassium nitrate will remain undissolved at this temperature.
Notice that you can increase potassium nitrate's solubility by increasing the temperature of the solution.
For example, increasing the temperature from #20^@"C"# to about #40^@"C"# would result in an increase in solubility from #"35 g"# to #"70 g"# per #"100 g"# of water.


