Question #933f1
1 Answer
Aug 14, 2016
Here's how I would do it.
Explanation:
1. Draw a cyclohexene ring.
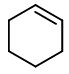
2. Attach a methyl group to one of the double-bonded carbons.
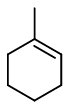
That carbon automatically becomes
The other double-bonded carbon becomes
3. Add
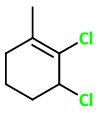
And you have the structure of 2,3-dichloro-1-methylcyclohexene.

