Question #25fae
1 Answer
Yellow-light photon has higher energy as compared to that of Orange-light photon.
Explanation:
Energy
where
We also know that for light, we have the following equation connecting velocity of light
Now look at the color constituents of white light as dispersed due to refraction
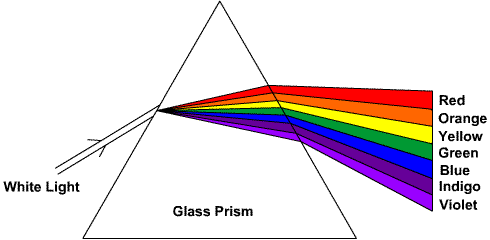
We remember sequence of these colors as VIBGYOR (as seen from bottom to top).
Violet color has the highest frequency and therefore, shortest wavelength at one end
Whereas Red has the lowest frequency and therefore, longest wavelength at the other end of spectrum.
Armed with knowledge we are ready to answer the question.
As seen from the spectrum, Yellow-light photon has higher frequency as compared to that of Orange-light photon.
Therefore, Yellow-light photon has higher energy as compared to that of Orange-light photon.
