Why does carbonate have a valence of two?
1 Answer
Nov 12, 2016
The valency of carbonate is two because it can combine with two hydrogen atoms.
Explanation:
Valency is the number of univalent atoms (e.g.,
The structure of carbonate is

Each
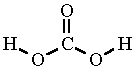
Since carbonate can combine with two

