What is the difference between molecular mass and formula unit mass?
1 Answer
Aug 14, 2017
The same as the difference between molecules and ionic compounds...
- Molecular mass is used for molecules, i.e. covalently-bonded multi-atomic substances.
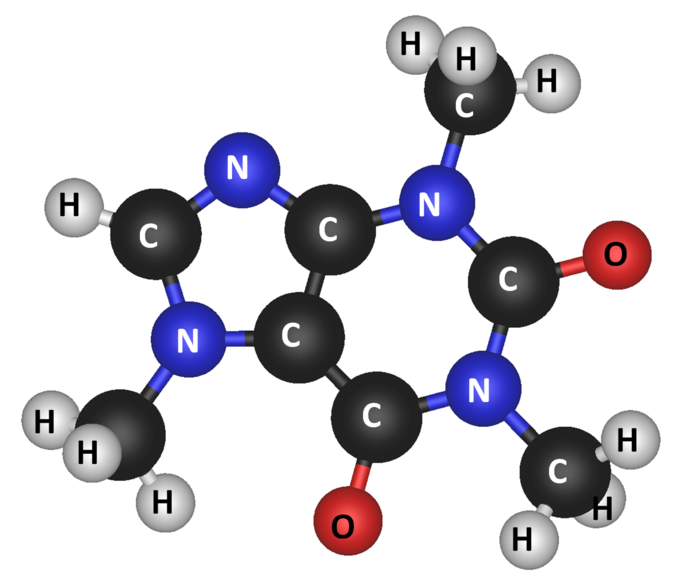
- Formula unit masses are used to describe ionic compounds, i.e. ionically-bonded compounds found as lattice structures. One formula unit of sodium chloride,
#"NaCl"# , is found in...
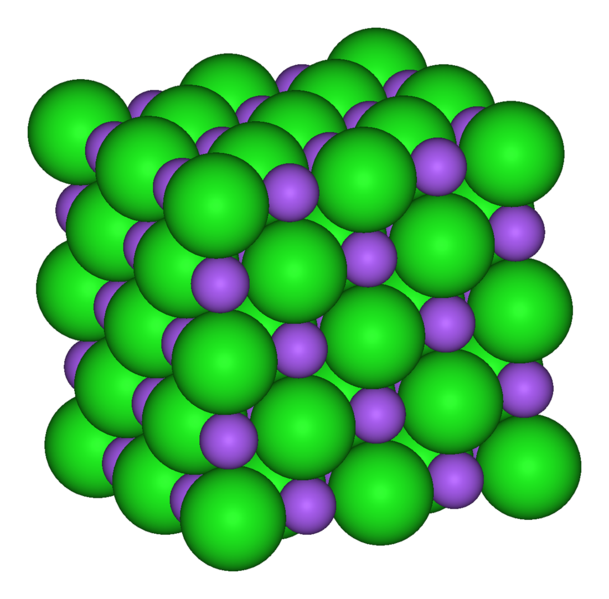
It makes no physical sense to prescribe a molecular mass to

