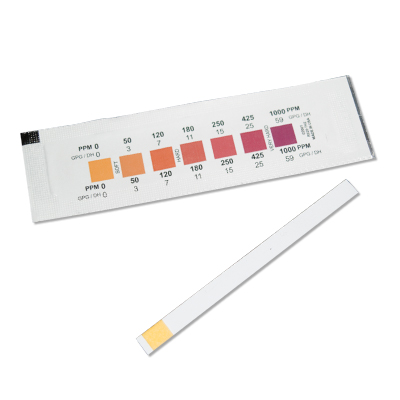
In water testing, paper strips often measure hardness in parts per million (ppm), where 1 ppm is defined as 1 mg of #"CaCO"_3# per litre of water.
Let's first calculate the concentration of #"Ca(OH)"_2# in parts per million.
#["Ca(OH)"_2] = (1000 color(red)(cancel(color(black)("g"))))/(1 color(red)(cancel(color(black)("m"^3)))) ×"1000 mg"/(1 color(red)(cancel(color(black)("g")))) × (1 color(red)(cancel(color(black)("m"^3))))/"1000 L" = "1000 mg/L" = "1000 ppm"#
The molar mass of #"Ca(OH)"_2# is 74.09 g/mol, and the molar mass of #"CaCO"_3#
is 100.09 g/mol.
Thus, we need 100.09 g of #"CaCO"_3# to get the same number of calcium ions as there in 74.09 g of #"Ca(OH)"_2#.
#"Hardness" = "1000 ppm" × 100.09/74.09 = "1350 ppm"#
This is VERY hard water. It would be off the scale on the test paper and give a deep purple colour.


