What considerations apply when a weak acid is titrated with a strong base, as opposed to a strong acid with a strong base?
2 Answers
We interrogate the equilibrium:
Explanation:
If, say, we have equimolar, say
When we titrate,
And thus, at equivalence,
The best demonstration of this is the behaviour in solution of
And thus the alkali metal fluorides give slightly basic solutions upon dissolution in water, due to the given equilibrium. On the other hand, bromide and chloride salts are the conjugate bases of STRONG acids, and their salts give neutral solutions.
In each case,
And of course, if you want
If you are unclear as to what I have argued, say so, and someone will readdress the problem.
The base is strong, so it reacts with the weak acid to the same extent as with the strong acid. The only difference between the weak and strong acid is how easily/quickly the neutralization occurs.
You will need to mix the combination of strong base and weak acid more thoroughly for longer, as the molecules will have a harder time finding each other, but it will occur with a similar volume of base added.
In other words, the thermodynamic result is the same (the yield is the same), but the kinetics of reacting a strong base with a weak acid are slower than the kinetics of reacting a strong base with a strong acid.
Here's an example titration graph of a strong base with weak acid:
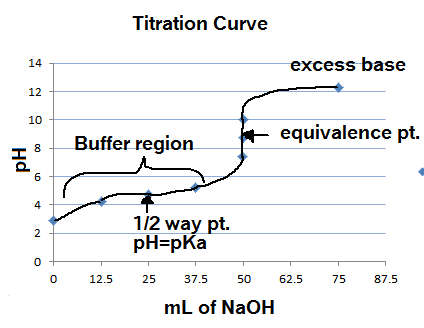 http://www.kentchemistry.com/
http://www.kentchemistry.com/
By Le Chatelier's principle, the strong base neutralizing some of the weak acid pushes the reaction to dissociate more of the weak acid until all of it is gone. That is how the above graph makes it all the way to the equivalence point, where all the acid was neutralized and its conjugate base is leftover.
Where is the half-equivalence point?
If you were to perform an experiment similar to the above and produced a graph for a strong base reacting with a monoprotic strong acid, the graph would be almost linear.
The weak acid solution has the highest
"HA"(aq) rightleftharpoons "H"^(+)(aq) + "A"^(-)(aq)
Smaller


