Question #de7c2
1 Answer
Apr 7, 2017
In a way it does, but it would be named differently.
Explanation:
Methoxy means
Methanal means
If we put this together we get:
Which is just the same as:
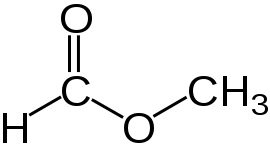
Which will be called methyl methanoate, because the C-atom that has the doubly bound O, also has the singly bound oxy group, and they combine to represent the -oate group.
It is also called methyl formate, being an ester of formic acid

