Are some metals soft, and volatile?
1 Answer
Well, some are, the majority are not............
Explanation:
Metals are NON-molecular materials. And the typical view of metals is
This view of metallic bonding rationalizes the physical properties of metals: (i) their
But look at some metals, for instance the alkali metals. These metals are SOFT, and exhibit very low melting and boiling points, especially for non-molecular materials. Mercury metal is even a LIQUID (one of the few elemental liquids).
Metals with several valence electrons TEND to be strong, high melting point materials. For instance iron, which as steel is the premier building material.
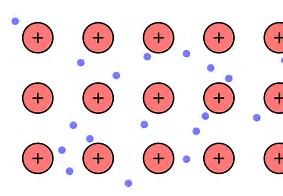
The diagram (if I have inserted it right!) depicts the electronic delocalization.

