Question #2de37
2 Answers
Te-137
Explanation:
U-235 Fission Rxn
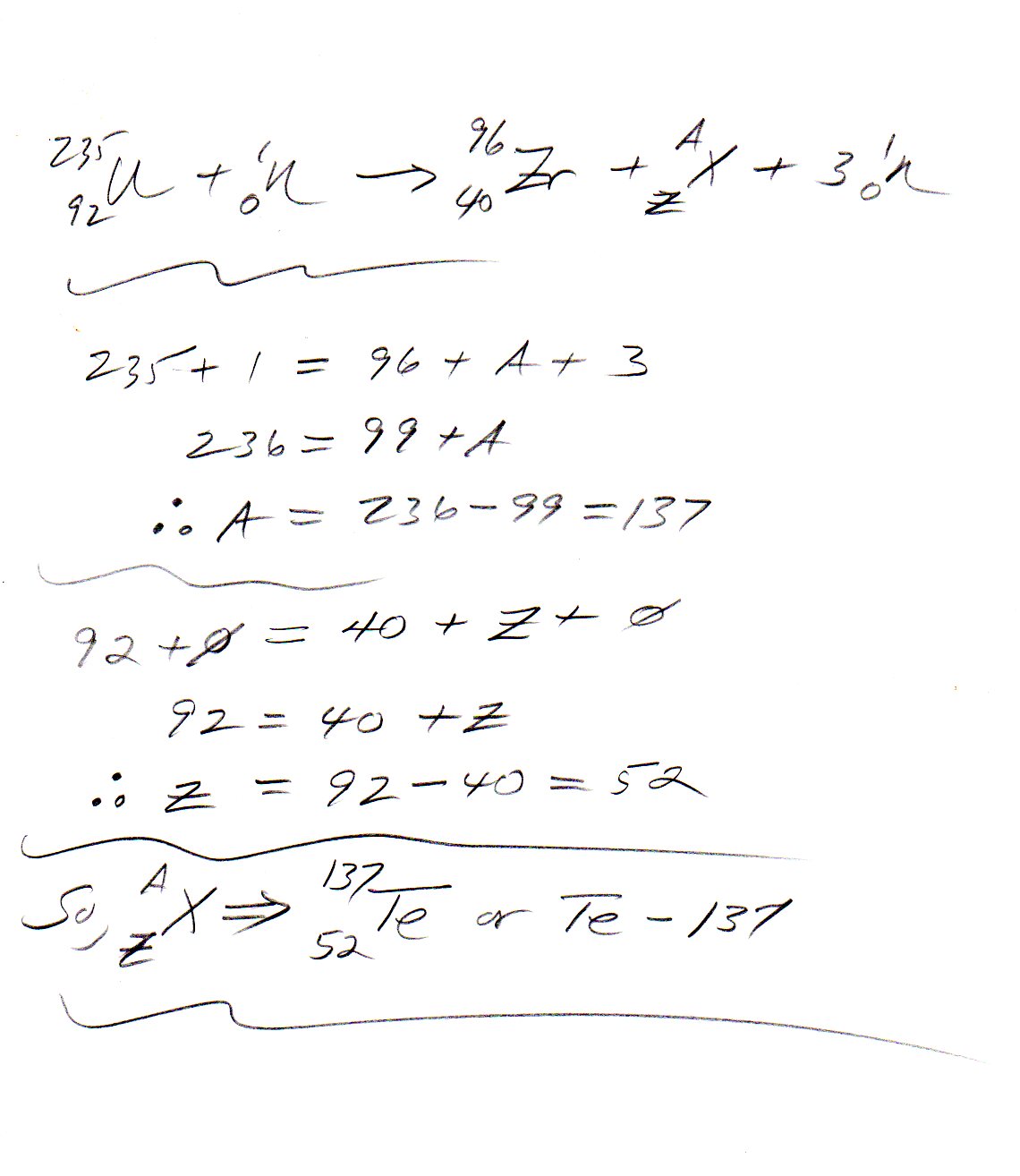
Zr-96 is an 'isotope' of Zirconium. That is, all Zirconium atoms in the universe have 40 protons (atomic number defines the element) but can have structures with different numbers of neutrons. By definition, isotopes are elements with the same atomic number but different atomic masses (due to variations in numbers of neutrons). Suggestion => Internet search for Zirconium Isotopes => Wikipedia to see the many isotopes of Zirconium. (https://en.wikipedia.org/wiki/Zirconium#Isotopes) Note, they all have 40 protons but due to variations in numbers of neutrons, there are multiple atomic masses. There are only 5 naturally occurring isotopes, but twenty-eight artificial isotopes of zirconium have been synthesized, ranging in atomic mass from 78 to 110
Also, the atomic mass of Zr published on most periodic tables is 91.224 amu (atomic mass units). This value is the weight average of the naturally occuring Zirconium isotopes that have been identified. So, Zirconium can have many isotopes (some natural and some synthetic) including Zr-96 (40 protons + 56 neutrons) & Zr-98 (40 protons + 58 neutrons). Nuclear fission is an example of synthetic Zirconium.
Te-137
Explanation:
Uranium-235 nucleus contains 92 protons and 143 neutrons.
- When bombarded with a neutron it becomes Uranium-236 which has 92 protons and 144 neutrons.
- On disintegration Uranium-236 yields two daughter nuclei and three neutrons.
- From Law of conservation of Charge the two daughters must be
(a) Given Zirconium-96 which has 40 protons and (b) X which must have#92-40=52# protons. We know that it must be Tellurium. - From Law of Conservation of mass we get
Atomic weight of Tellurium nucleus#X# as
#236=96+3xx1+"Te"#
#236=96+3+"Te"#
#=>"Te"=137#
Nuclear reactions can be written as
Bombardment
#"U"_92^235+n_0^1->"U"_92^236#
Disintegration
#"U"_92^236->3n_0^1+"Zr"_40^96+"Te"_52^137#

