Question #1f162
1 Answer
Aug 1, 2017
No.
Explanation:
Let's looks at the structure of acetic acid.
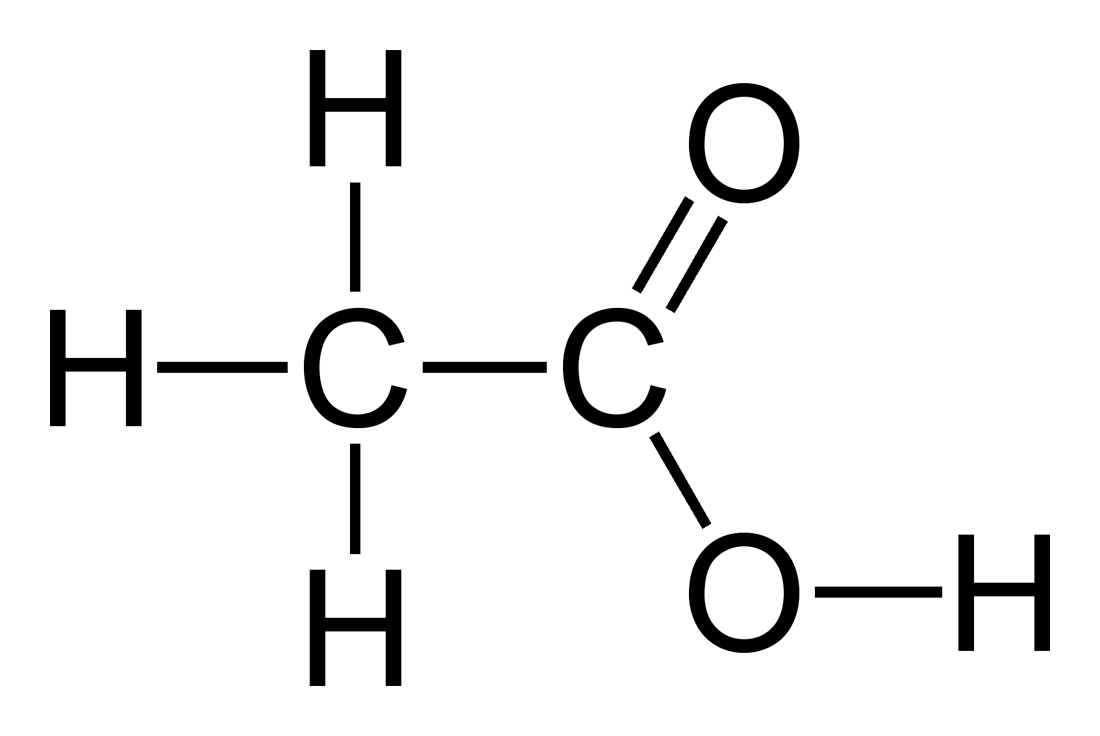 https://en.wikipedia.org/wiki/Acetic_acid
https://en.wikipedia.org/wiki/Acetic_acid
The carbonyl oxygen is double bonded to the carbonyl carbon. Double bonds are stronger than single bonds as share more electrons between two atoms. Since oxygen is more electronegative than carbon, electrons will impart a more negative charge on oxygen.
If you look at the structure of acetic acid, the oxygens will have partial negative charges. The carbonyl oxygen will have a greater negative charge than the other oxygen. The hydrogens and the carbonyl carbon will have a partial positive charge. The methyl carbon may have a slight negative charge since there are three hydrogens bonded to it.

