Question #d4e7e
1 Answer
Sep 6, 2017
1- Gas particles are separated with no regular arrangement.
2- Liquid particles close together with no regular arrangement.
3- Solid particles are tightly packed, usually in a regular pattern.
Explanation:
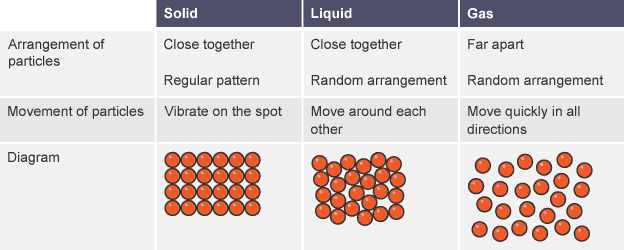
Gas molecules moves freely and have empty spaces that is why they can be compresses, so gas has no definite shape nor definite volume.
Liquid particles are in cluster form and move freely, there is very little space among these cluster ,so these cannot be compressed at nominal pressure, liquid has no definite shape, but has definite volume.
Particles of solid are tightly packed cannot move at all, just vibrate about their axis, so solid have definite shape and definite volume.
Image BBC Bitesize

