Question #694c2
1 Answer
I'd go with option (1).
Explanation:
In order for a weak base-conjugate acid buffer to resist the addition of equal amounts of a strong acid or a strong base, respectively, it must contain equal concentrations of the weak base and of its conjugate acid.
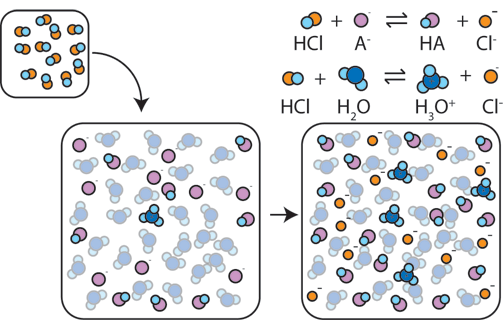
A buffer resists significant changes in
In your case, the concentration of the weak base exceeds the concentration of the conjugate acid, so the buffer will be able to resist the addition of a greater amount of strong acid than of strong base.
In other words, the conjugate acid will be consumed faster than the weak base when equal amounts of a strong acid or of a strong base are used.
This means that its anti-base ability will be lower than its anti-acid ability, so the buffer will have
#"anti-acid ability " > " anti-base ability"#
If you use the Henderson - Hasselbalch equation for a weak base-conjugate acid buffer as reference
#"pH" = 14 - overbrace(["p"K_b + log( (["NH"_4^(+)])/(["NH"_3]))])^(color(blue)("= pOH"))#
you can say that when
#["NH"_3] > ["NH"_4^(+)]#
you have
#log( (["NH"_4^(+)])/(["NH"_3])) < 0#
which implies that the
#["NH"_3] = ["NH"_4^(+)] => "pH" = 14 - "p"K_b#
#["NH"_3] > ["NH"_4^(+)] => "pH" > 14 - "p"K_b#
Now, we usually consider the buffer to be effective at a
#Delta_ ("pH") = (14 - "p"K_b) +-1#
So for a weak base - conjugate acid buffer, you have a
#Delta_ ("pH") = [(13 - "p"K_b)" , " (15 - "p"K_b)]#
If you start with a buffer that has
#["NH"_3] > ["NH"_4^(+)]#
its
# ["NH"_4^(+)] > ["NH"_3] -> "the pH is closer to "(13 - "p"K_b)# #["NH"_4^(+)] = ["NH"_3] -> "the pH is equal to " (14 - "p"K_b)# #["NH"_4^(+)] < ["NH"_3] -> "the pH is closer to " (15 - "p"K_b)#
This means that a smaller quantity of strong base will be needed to increase its
This is why you can say that the buffer has
#"anti-acid ability " > " anti-base ability"#

