Question #136e3
1 Answer
Helium.
Explanation:
This question is actually asking about atomic radius, which is the average distance between the nucleus to the boundary electron cloud (which are the valence electrons).
The more attracted the valence electrons are to the nucleus, the smaller their atomic radius.
This attraction, or atomic radius, is influenced mainly by how positive the nucleus is, and how far away it is from the nucleus.
For example, valence electrons in lithium are less attracted to their nucleus than valence electrons in fluorine are.
This is because fluorine both has more protons than lithium, making for a more positive nucleus.
Valence electrons in fluorine also experience a greater attraction to the nucleus than valence electrons in potassium.
This is because, while potassium has a more positive nucleus due to more protons, the increased attraction is outweighed by the sheer number of energy levels it has. This increases distance and decreases attraction drastically.

Therefore, we can conclude that:
- Atomic radius increases from right to left.
- Atomic radius increases from up to down.
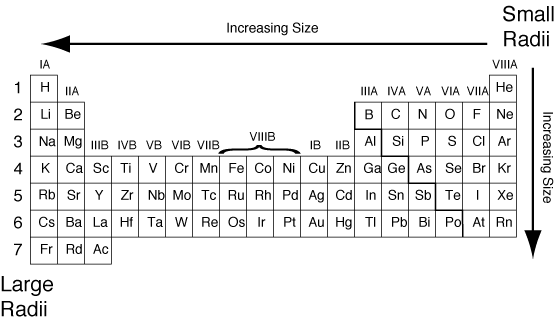
Helium is in the upper right corner, so it will have the smallest atomic radius and hence the greatest attraction between its valence electrons and nucleus. :)

