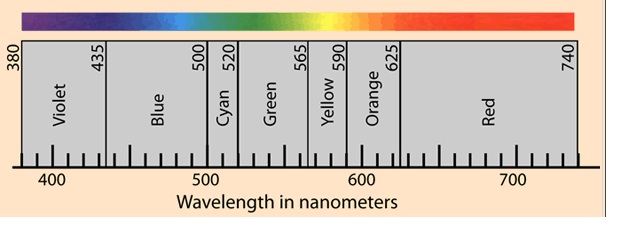
A beam of light has a wavelength of 506 nanometers. What is the frequency of the light? What color is the light?
1 Answer
Feb 18, 2016
I found:
Explanation:
You can find the frequency
where:
So:
It should be Cyan (