A sample of carbon is placed in a rigid 1.50 L flask. Dinitrogen monoxide is added and the flask heated to 800C. When the system reached equilibrium the partial pressure of the carbon dioxide is found to be 0.030 atm and the partial pressure of the dinitrogen monoxide to be 1.48 atm. What is the Gibbs free energy change, delta G, for the reaction at 800C?
1 Answer
Apr 27, 2015
The value of
I presume that the reaction is
Step 1. Calculate
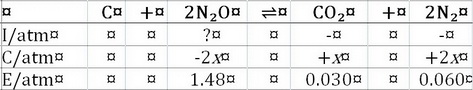
From the ICE table, we see that
So
Step 2. Calculate

