An unknown solution changed the color of a phenolphthalein indicator from coloriess to pink. What would you comment about the pH of the unknown solution?
1 Answer
May 11, 2017
That you added a basic solution, i.e. .............
Explanation:
.....a solution whose
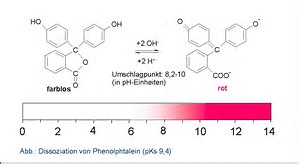
I found that phenolpthalein was one of the very best indicators to use for standard acid-base titrations, in that the acidic form is colourless,

