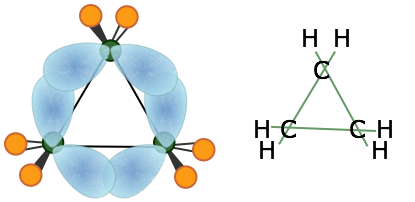Are epoxides significantly more reactive than simple ethers? Why?
1 Answer
May 8, 2015
Yes. They have much more steric strain and are therefore more susceptible to nucleophilic attack. They are essentially cyclopropane rings with one oxygen replacing one carbon. Cyclopropane rings have "banana" bonds for the sake of improving stability, but that doesn't really improve stability that much.