Do metals have localized electrons?
1 Answer
Oct 22, 2016
Yes they do.
Explanation:
Metal atoms contain electrons in their orbitals. When they undergo metallic bonding, only the electrons on the valent shell become delocalized or detached to form cations. The atoms still contain electrons that are 'localized', but just not on the valent shell.
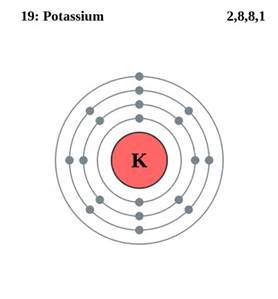
The electron on the outermost shell becomes delocalized and enters the 'sea' of delocalized electrons within the metal lattice. The electrons on the other shells are unaffected so remain attached to the atom.

