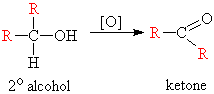Do pi bonds change the shape of a molecule?
1 Answer
Your question is vague but I will try to answer
When determining the shape of a molecule, one must count the electron domains around the central atom. An electron domain is considered to be any bond (single, double, triple) or a non-bonding pair of electrons.
Pi bonds are the additional orbital overlaps that make up double and triple bonds.
If you are looking at a molecule like the carbonate ion, there are 3 electron domains and the molecule shape is trigonal planar - the pi bond in the double bond has the same effect on the shape as a single (or triple bond)
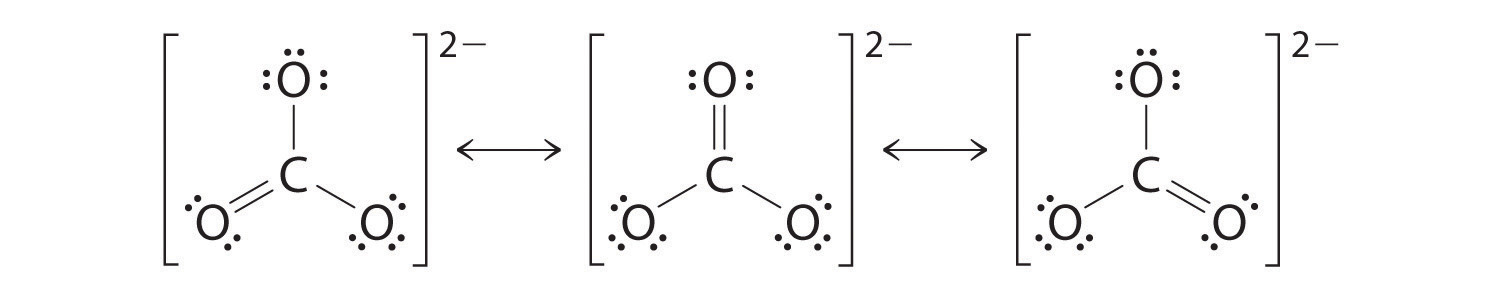
HOWEVER
If you are looking at the formation of a pi bond in a chemical reaction, a new molecule is formed and it could have a different shape from the reactant molecule (for example, oxidizing a secondary alcohol to a ketone - changes the shape from tetrahedral to trigonal planar)