How are alkyl groups electron donating?
1 Answer
Carbon is more electronegative than hydrogen; therefore, its tendency to donate electrons as part of an alkyl group is increased.
Explanation:
Recall that an isolated alkyl group has a carbon atom at one end / point that has not completed its valence shell (this disobeys the octet rule, with carbon being one of the few elements that reliably obeys it). This means that there are electrons in its valence shell that have not participated in covalent bonding. The alkyl group's tendency to donate electrons derives from this fact.
Electronegativity is defined as the tendency of an atom of an element to draw electrons towards itself in a covalent bond. Electronegativity is a property of an element, and it is the arbitrary Pauling scale that represents how electronegative each element is.
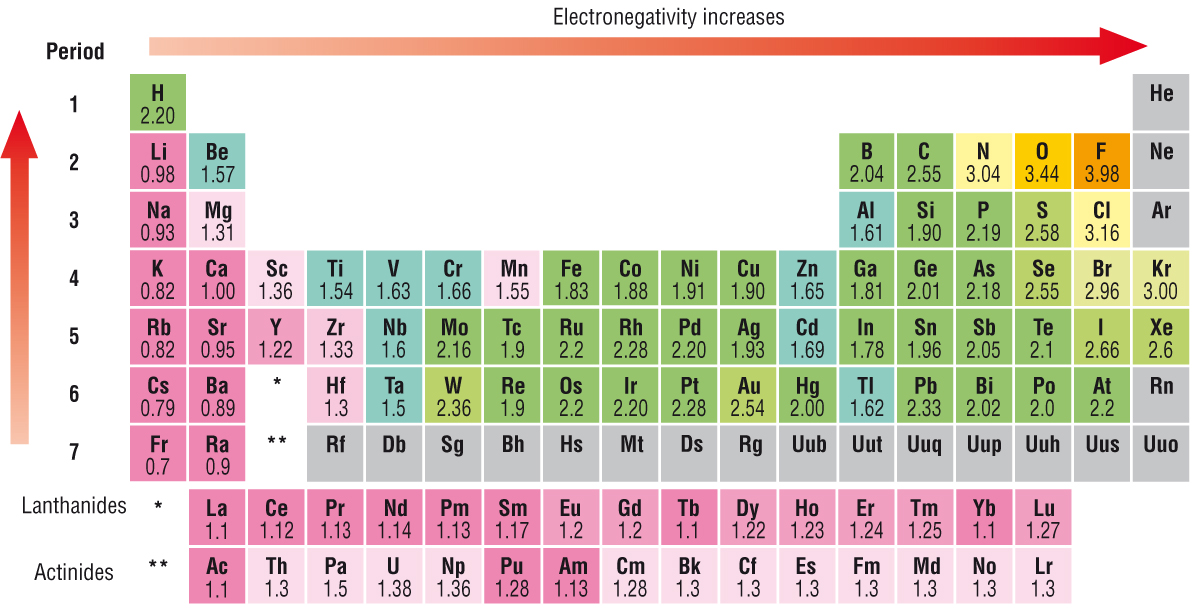
See how hydrogen has been assigned the value
This leads to the carbon atom having an electron excess, and so a partially negative charge (
As a result of the carbon atom's
So how does this change with carbon chain length in the alkyl group? As an example, the effect of this is greater in an ethyl group than in a methyl group, because the

