How can I draw 1,2,3,4,5,6-hexachlorocyclohexane with all the chloro groups in equatorial positions?
1 Answer
There are two kinds of substituents that can be attached to a chair conformation for a cyclohexane - axial substituents and equatorial substituents.
The axial substituents are drawn pointing straight up and straight down on the chair conformation, while the equatorial substituents are drawn pointing out to the sides of the chair. For cyclohexane, or
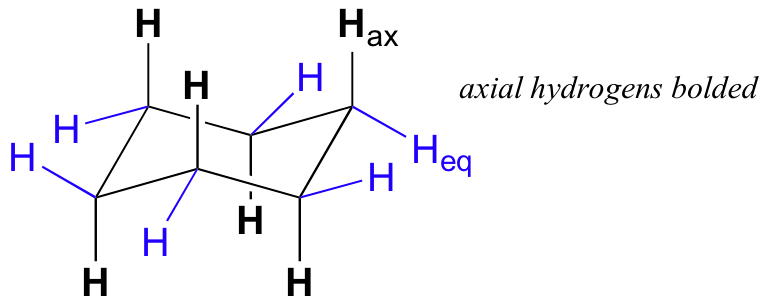
In the above picture, the equatorial hydrogens are drawn in blue. If you look closely, you'll see that these equatorial hydrogens alternate between UP and DOWN positions on the chair.
UP is a term used to describe groups that are coming out of the plane of the page (are placed on a wedge), while DOWN is used to describe groups that are going into the plane of the page (are placed on a dash).
Hexachlorocyclohexane is
If you start by looking at the carbon atom labeled (1), and go clockwise through all the equatorial substituents, you'll have: DOWN, UP, DOWN, UP, DOWN, and finally UP (the chlorine attached to the carbon labeled (6)).
In the same order, the chlorine atoms will be placed on a dash (carbon (1)), on a wedge, on a dash, on a wedge, on a dash, and finally on a wedge (carbon (6)). The wedge-dash notation for the compound would look like this

