How can I draw activation energy in a diagram?
1 Answer
Jun 26, 2015
You follow a series of steps.
Explanation:
1. Draw and label a pair of axes.
Label the vertical axis "Potential Energy" and the horizontal axis "Reaction Coordinate".
2. Draw and label two short horizontal lines to mark the energies of the reactants and products.
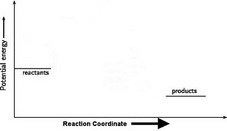
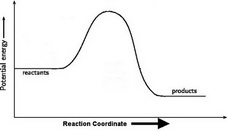
3. Draw the energy level diagram.
There must be a hump in the curve to represent the energy level of the activated complex.
4. Draw and label the activation energy.
Draw a horizontal line from the highest part of the curve towards the vertical axis.
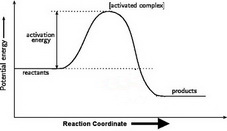
The activation energy

