How can metals be distinguished from non metals on the periodic table?
1 Answer
Unfortunately, there's no universal agreement on this, but many versions of the Periodic Table will offer some guidance.
Explanation:
Certainly, all of the elements in Groups 1-12 except hydrogen are considered metals.
There's pretty good agreement that all of the elements in columns 17 and 18, plus elements
As you go down a Group, the elements become more metallic.
Most chemists would say that
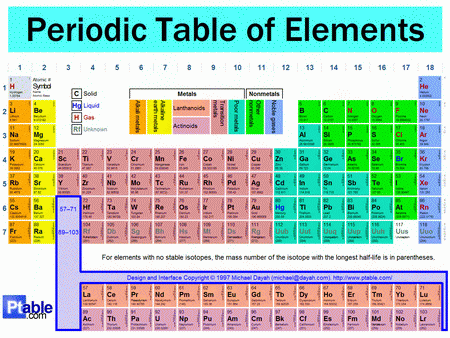
(From www.ptable.com)
In between these broad groups are the metalloids and semiconductors, and different versions of the Periodic Table differ in how individual elements in this region are classified.


