How can rate of reaction be affected?
1 Answer
Several factors can influence the rate of a chemical reaction. In general, anything that increases the number of collisions between particles will increase the reaction rate, and anything that decreases the number of collisions between particles will decrease the chemical reaction rate.
NATURE OF THE REACTANTS
In order for a reaction to occur, there must be a collision between the reactants at the reactive site of the molecule. The larger and more complex the reactant molecules, the less chance there is of a collision at the reactive site.
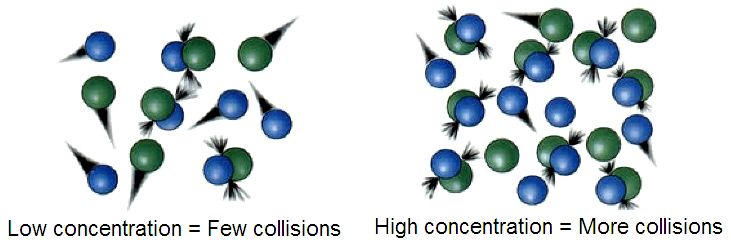
CONCENTRATION OF REACTANTS
A higher concentration of reactants leads to more effective collisions per unit time and leads to an increasing reaction rate.
PRESSURE OF GASEOUS REACTANTS
Changing the pressure of gaseous reactants is, in effect, changing their concentration. The increased number of collisions caused by a higher pressure generally increases the reaction rate.
PARTICLE SIZE OF SOLID REACTANTS
Reaction depends on collisions. If a reactant is a solid, grinding it into smaller particles will increase the surface area. The more surface area on which collisions can occur, the faster the reaction.
TEMPERATURE
Usually, an increase in temperature causes an increase in the reaction rate. A higher temperature means that the molecules have a higher average kinetic energy and more collisions per unit time. It also increases the number of collisions that have enough energy to cause a reaction to take place.
MEDIUM
The rate of a chemical reaction depends on the medium in which the reaction occurs. It may make a difference whether a medium is aqueous or organic; polar or nonpolar; or solid, liquid, or gas.
CATALYSTS
Catalysts lower the activation energy of a chemical reaction and increase the rate of a chemical reaction without being consumed in the process. The do this by an alternative mechanism that has a lower activation energy.