How do hypertonic solutions effect plant cells?
1 Answer
Hypertonic solutions make plant cells lose water.
Explanation:
Hypertonic solutions have a higher solute concentration. When plant cells are placed in such solutions, water will move from inside the plant cell to the outside of the cell, resulting in the shrinking of the cell (the cell is said to be plasmolyzed).
This occurs because of osmosis. When there are solutes on two sides of a membrane, a balance of solute on the two sides of the membrane will be attempted. The molecules on both sides of the membrane will try to move across the membrane, but the net movement will be down the concentration gradient (from high to low concentration).
In a hypertonic solution, there is less water outside than inside the plant cell, so the water within the plant will try to diffuse outside in order to achieve equilibrium.
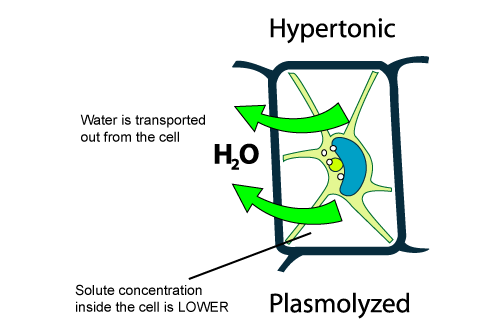
This video explains the changes that occur in cells in both hypertonic and hypotonic solutions.
This video shows onion cells losing water because of osmosis. The slide was originally made using distilled water, then drops of concentrated salt were added to the edge of the cover slip. The salt diffuses under the cover slip and plasmolysis begins at 0:48.


