How do solutes and solvents interact?
1 Answer
Mar 2, 2016
Explanation:
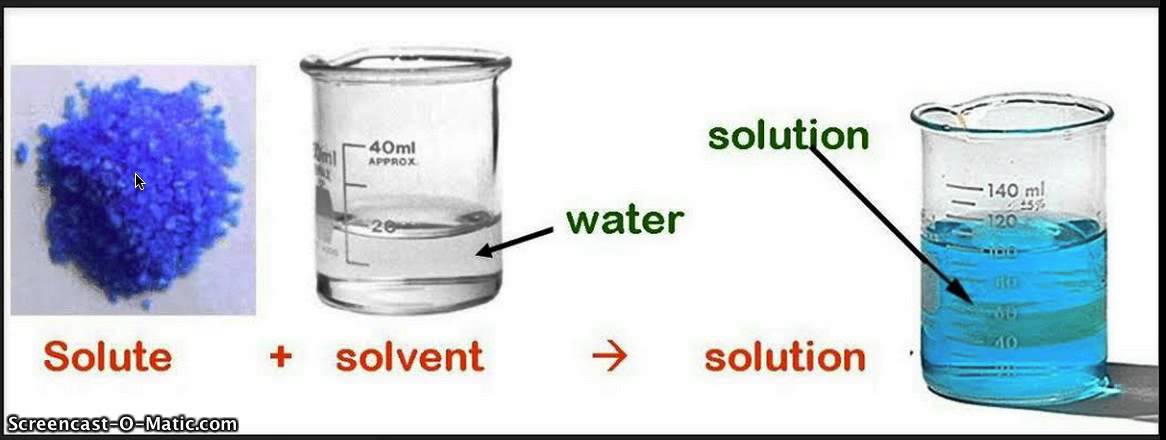
When a solute is placed in a solvent, the concentrated solute begins to slowly break apart into tiny pieces.
The molecules of the solvent began to move out of the way in order to make room for the molecules of solute.
This interaction continues until both the solute and the solvent has equal concentration throughout.
 Screencast-O-Matic.com
Screencast-O-Matic.com

