How does osmosis relate to solute concentration?
1 Answer
Nov 22, 2014
Osmosis is the net movement of a solvent, usually water, across a semi-permeable membrane from where it is in higher concentration to where it is lower in concentration.
Explanation:
Osmosis relates to solute concentration in that when solute concentration is lower, the concentration of solvent is higher, and when the solute concentration is higher, the concentration of solvent is lower.
So, we could also say that osmosis is the net movement of a solvent across a semi-permeable membrane from an area of low solute concentration to an area of greater solute concentration.
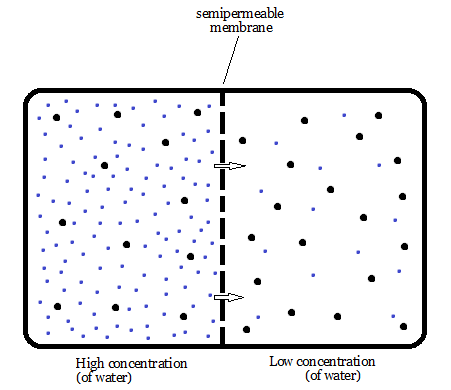
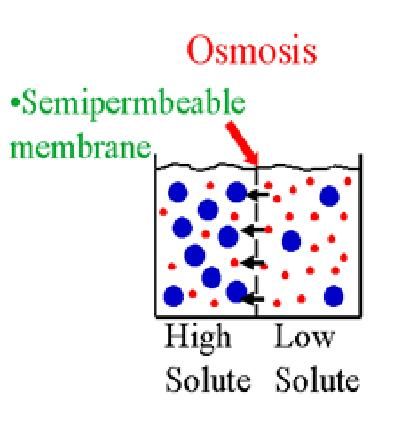
Here is a video which discusses changes that occur in plant cells when they are placed into hypertonic and hypotonic solutions.


