How do you balance #Ni + C_4H_8N_2O_2 -> Ni(C_4H_8N_2O_2)_2#?
2 Answers
Explanation:
as there are 1 molecule nickel, 4 molecules of carbon, 8 molecules of hydrogen, 2 molecules of nitrogen, and 2 molecules of oxygen in left hand side
but 1 molecule of nickel, 8 molecules of carbon, 16 molecules of hydrogen, 4 molecules of nitrogen, and 4 molecules of oxygen
so, by multiplying
Explanation:
The starting chemical equation given to you is actually incorrect.
This reaction takes place in aqueous solution and it involves the nickel(II) cation,
Reacting nickel(II) cations with dimethylglyoxime,
The nickel(II) cations can be delivered to the solution by a soluble salt like nickel(II) nitrate,
The actual balanced chemical equation for this reaction can be written like this
#"Ni"_text((aq])^(2+) + 2"C"_4"H"_8"N"_2"O"_text(2(aq]) -> "Ni"("C"_4"H"_7"N"_2"O"_2)_text(2(s]) darr + 2"H"_text((aq])^(+)#
The reaction involves two dimethylglyoxime molecules acting as chelating agents to form the nickel dymethylglyoxime complex.
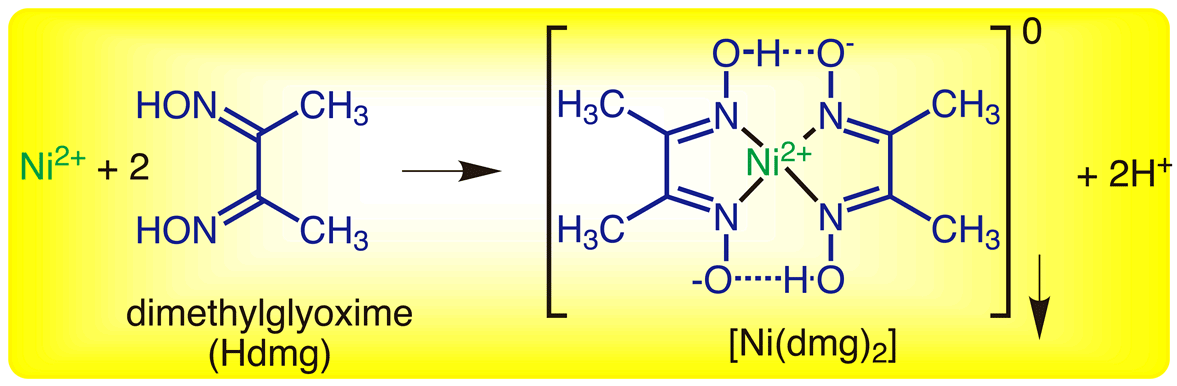
When the nickel(II) cations are delivered via nickel(II) nitrate, the reaction will produce
#"Ni"("NO"_3)_text(2(aq]) + 2"C"_4"H"_8"N"_2"O"_text(2(aq]) -> "Ni"("C"_4"H"_7"N"_2"O"_2)_text(2(s]) darr + 2"HNO"_text(3(aq])#
The nitrate anions,
This reaction is used as a confirmation test for the presence of nickel(II) cations. Nickel dymethylglyoxime is a red precipitate.



