How do you draw and name all isomers of #C_3H_4Cl_2#?
1 Answer
Sep 22, 2016
You first draw the parent compound propene
Explanation:
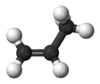
There is a double bond in it:
Now replace two of the
Then there's another parent compound
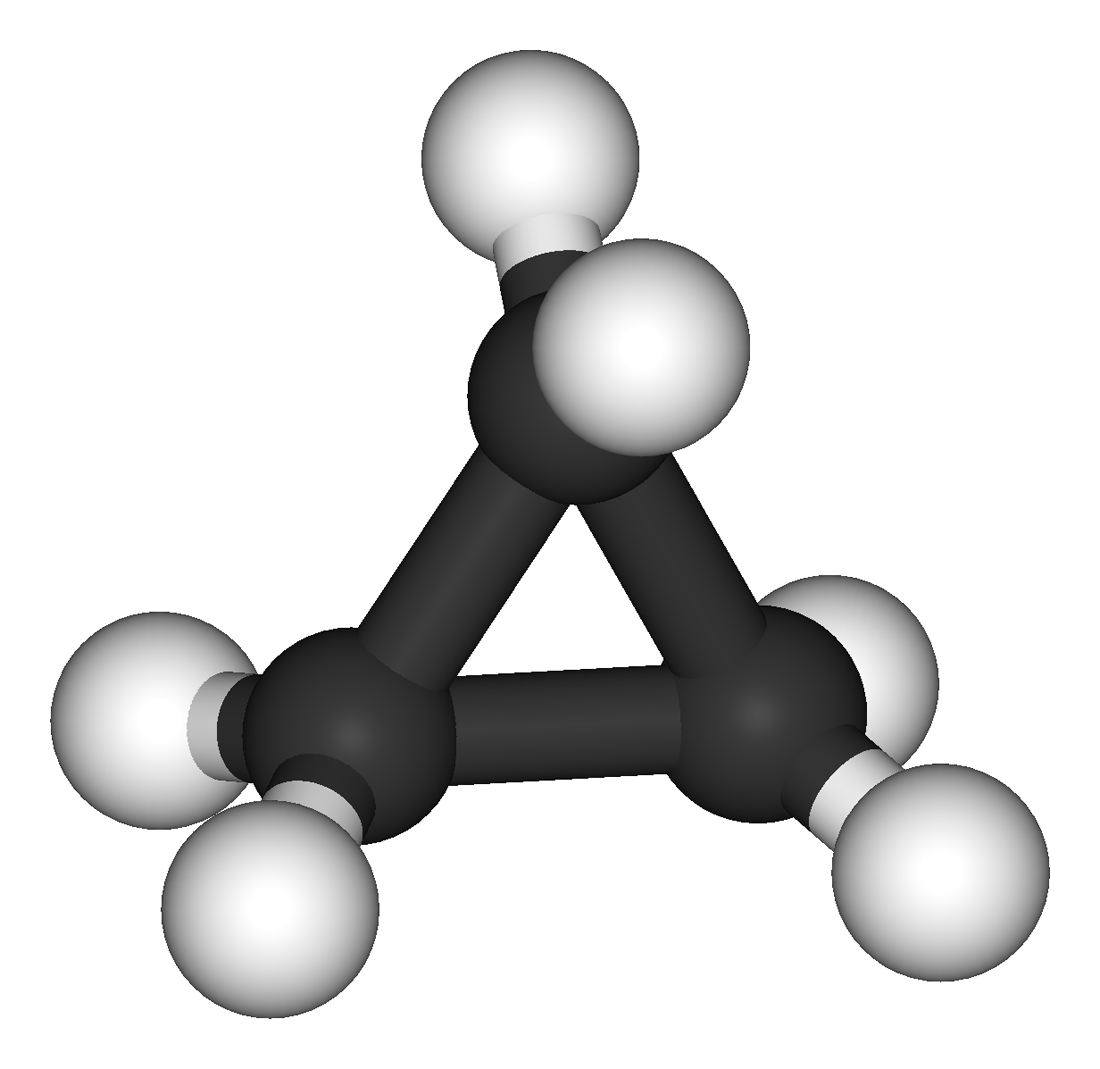
Here there are only two possibilities to replace two
Both on the same
On two neighbouring

