How do you read the phase diagram of water?
1 Answer
Well, first you need a phase diagram........and then you must decide on conditions of temperature and pressure.
Explanation:
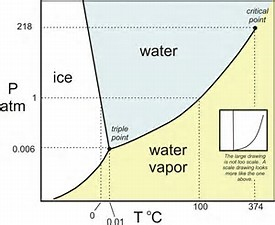
Typically, pressure is plotted against temperature, and here we examine the phase of the water under different conditions of temperature and pressure. Of course, we can draw the ordinate
The fact that the slope of the ice-water transition is NEGATIVE is a very unusual property, and it can show that ice is less DENSE than water (I think the slope of the graph is highly exaggerated!).
Depicted on the graph are the

