How does atomic size vary on the periodic table?
1 Answer
Explanation:
Atomic size decreases across a Period from left to right as we face the Table, but INCREASES down a Group, a column of the Periodic Table. This is one of the most fundamental and useful ideas you can learn as you are introduced to chemistry.
Consider the diagram,
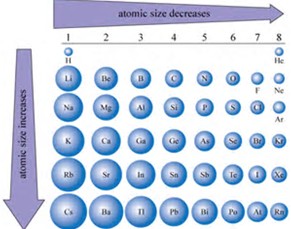
Atomic size is influenced by (i) nuclear charge,
Once a valence shell is filled, the nuclear charge is shielded with reasonable effect. A new quantum shell is initiated, farther from the nuclear core, with the Periodic pattern again repeated across the new Period.
I urge you to read your text in this respect. This contest between nuclear charge,

