How does concentration affect galvanic cell?
1 Answer
The Anodic Cell should start with a very low concentration of Reducing Agent ion and the Cathodic Cell should start with a high concentration of Oxidizing Agent ion. Such is the general rational for design of a Galvanic Cell System.
Explanation:
To optimize the voltage in a Galvanic/Voltaic Cell, the concentration of the Reducing Agent in the Anodic Side of the system (oxidation rxn) should be low and the concentration of the Oxidizing Agent ion in the Cathodic side of the system (reduction rxn) should be high. As the system discharges the concentration of Reducing Agent ion will increase due to oxidation process in the Anodic Cell and the concentration of Oxidizing Agent ion will decrease due to reduction process in the Cathodic Cell of the system.
A plot of voltage vs time for a typical Voltaic Cell demonstrates a 1st order type of decay trend as shown in figure 1 below. The Nernst Equation Supports the annotations listed for concentrations of oxidizing and reducing agents at specified time of decay.
Figure 1: Decay Curve for Voltaic Cell
(Graphic, Doc048, 8/2017)
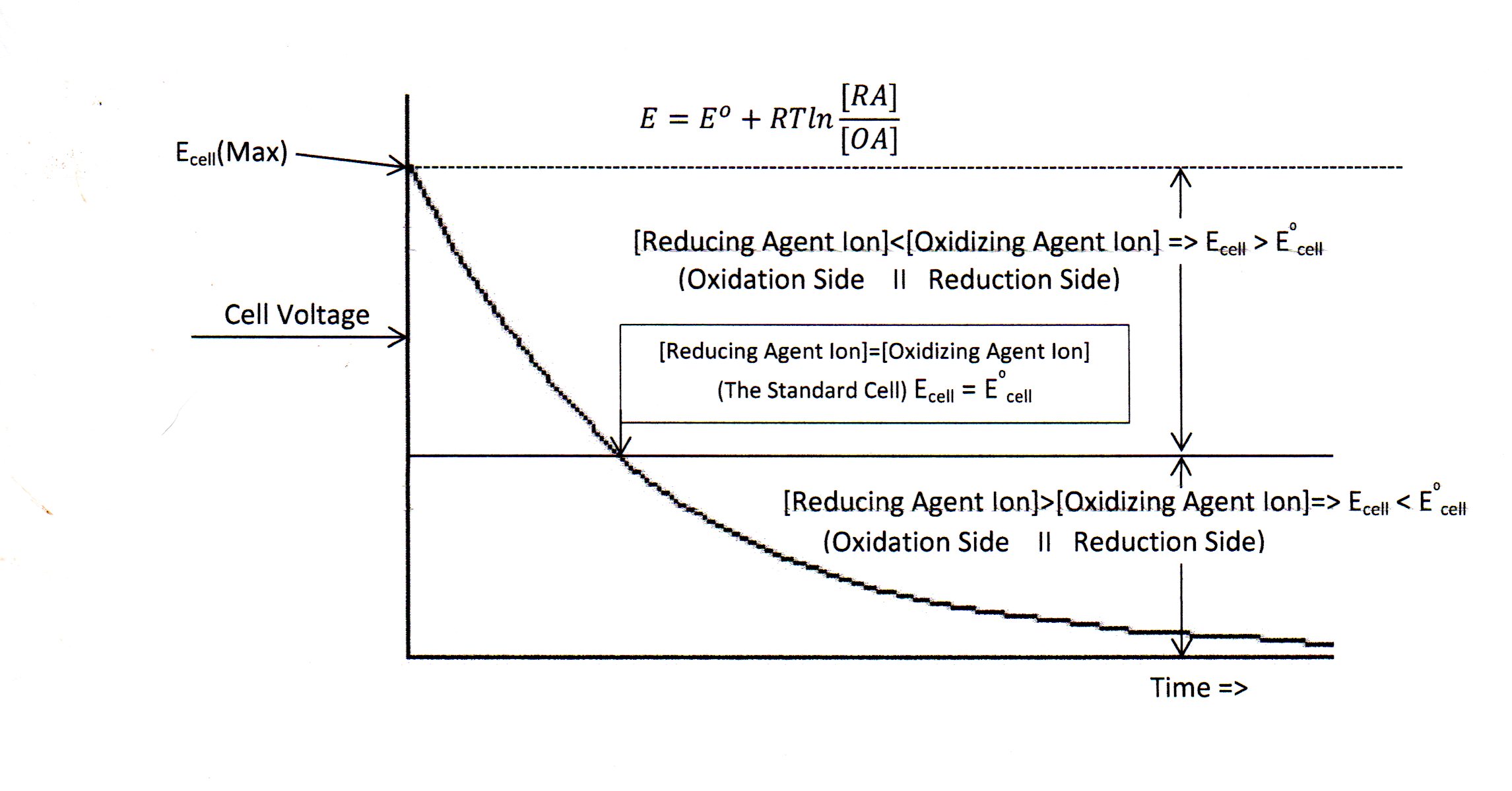
For oxidation side ion concentration [Reducing Agent] less than the reduction side ion concentration [Oxidizing Agent] the cell voltage will be above the standard cell value, but when oxidation side ion concentration [RA] becomes greater than reduction side ion concentration [OA] the cell voltage will be less than the standard cell. When the ion concentrations on the oxidation and reduction sides of the cell are equal ([RA] = [OA]), then standard cell voltage occurs.

