Galvanic Cells
Key Questions
-
Answer:
An electrochemical cell which produces an electric current from a redox reaction.
Explanation:
Let's look at the following redox reaction:
#Zn + Cu^(2+) -> Zn^(2+) + Cu# From the oxidation states, we know that
#2# electrons are transferred from#Zn# to#Cu^(2+)# .When this redox reaction happens directly, the electrons are also transferred directly—which isn't helpful if we want to conduct an electric current.
Galvanic cells usually fix this problem by separating the reactants into two half-cells and connecting them through a wire.
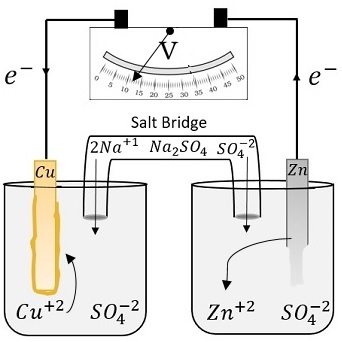
When
#Zn# and#Cu^(2+)# are separated, electrons will flow from#Zn# through the wire to reach#Cu^(2+)# .
This flow of electrons will create an electric current, which can be used to do work!The two half-cells in a galvanic cell are the cathode and anode.
- The cathode is the half-cell which is receiving electrons.
For us, it's the copper half-cell. - The anode is the half-cell which is giving electrons.
For us, it's the zinc half-cell.
It's also important to note that there is a salt bridge which allows ions to flow to the two half-cells.
If we didn't have a salt bridge, we'd see a build-up of negative charge in the cathode and a build-up of positive charge in the anode. If this happens, electrons won't be able to flow from the anode to the cathode anymore.
Salt bridges solve this issue by allowing positive ions like
#Na^+# to flow into the cathode and negative ions like#SO_4^(2-)# to flow into the anode, maintaining charge. - The cathode is the half-cell which is receiving electrons.