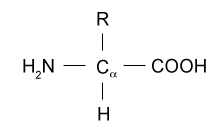
How does pH affect amino acid structure?
1 Answer
Protonation and deprotonation of the amino acid
Explanation:
The pH of an amino acid affects which atoms protonate and deprotonate. Let's start by looking at the generic structure of an amino acid.
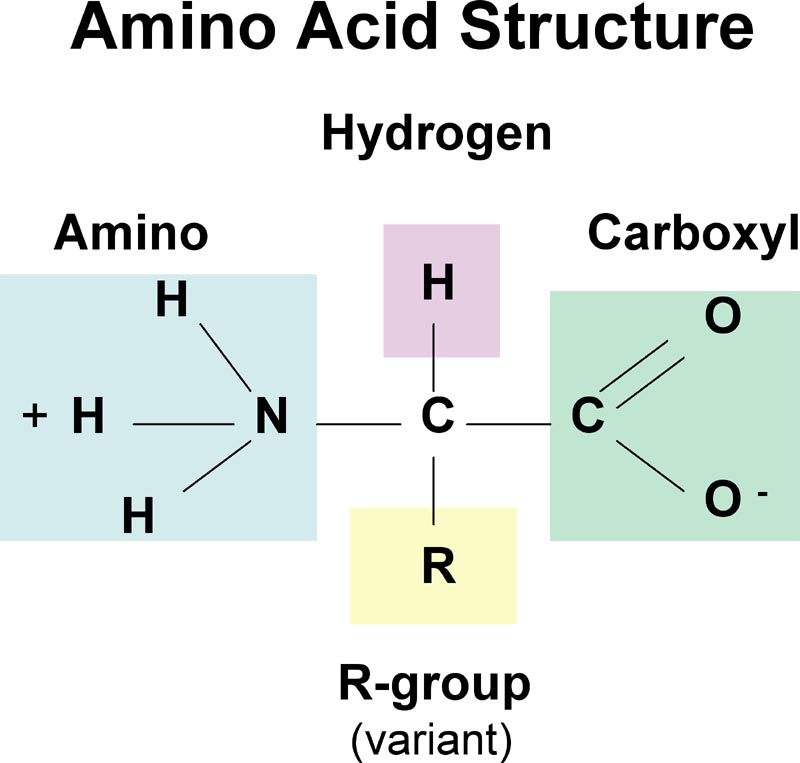
Notice something weird? The amino group is protonated but the carboxyl is not. Amino acids are amphoteric, meaning they can act like an acid and base. Also, amino acids are dipolar. Amphoteric, dipolar species are called zwitterions.
This is due to ammonium (amino) groups being less acidic than carboxylic acids. Look at the
For the amino acids with protonated R groups, you need to pay attention to their
The exact opposite would happen for protonation of amino acids. The
Here is the structures at increasing pHs for a generic amino acid. If you start at a high pH and decrease it, read the structures from right to left.
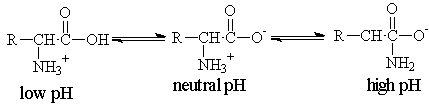
Here is an example of deprotonating a protonated R group (glutamic acid).
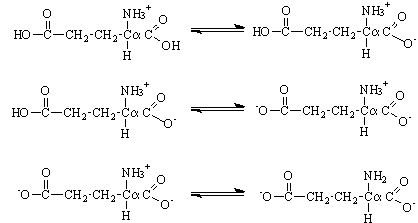
Note that this is the WRONG structure at a neutral pH. This is usually a trap answer on tests.