How does pH change when a strong acid is titrated with a strong alkali?
1 Answer
Jun 4, 2018
From
Explanation:
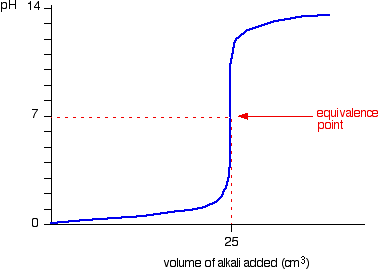
Strong alkali added to the acid to for the following titration curve
There's a sharp

