How does phenol differ from benzene?
2 Answers
Explanation:
Both Benzene and Phenol are aromatic compounds.
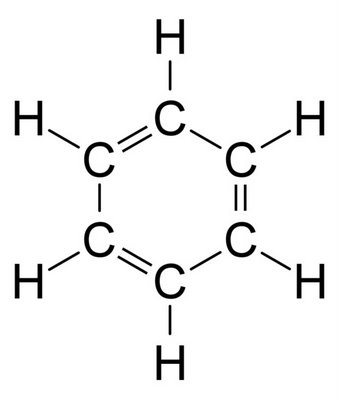
Benzene:-
- molecular formula of
#C_6H_6# - Neutral in nature and mostly acts as a solvent
- Possess a sweet odor.
- Colorless liquid
- Less polar in nature than phenol
- Less soluble in water than phenol
- Faster evaporation.
 wikipedia.org
wikipedia.org
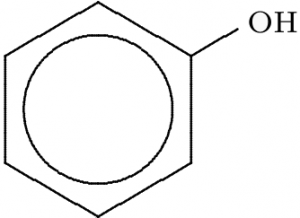
Phenol:-
- molecular formula
#C_6H_6OH# - Acidic in nature
- Possess a strong odor.
- Phenol in solid form is white crystalline color.
- More polar in nature than benzene.
- Easily soluble in water
- Slower evaporation.
 wikipedia.org
wikipedia.org
Simply by the presence of the hydroxyl functional group...
Explanation:
And of course the hydroxyl group confers vastly different chemical and physical properties.
We got benzene,
This difference in volatilities relates to the degree of
And why should phenol have a greater intermolecular force? Well, due to the presence of the hydroxyl group that offers the possibility of
And chemically, the hydroxyl substitution makes phenol a much more reactive substrate in the context of aromatic electrophilic substitution. To achieve this for the benzene ring, often some Lewis-acidic catalyst has to be added, before the ring reacts with an electrophile. For phenol, OFTEN, this catalyst may be omitted, and substitution occurs facilely.


