How many minutes will it take for an isotope that has a half life of 45 minutes to decay to 1/8th of its original amount?
1 Answer
Jul 3, 2014
Half-life (t½) is the amount of time required for a quantity to fall to half its value as measured at the beginning of the time period.
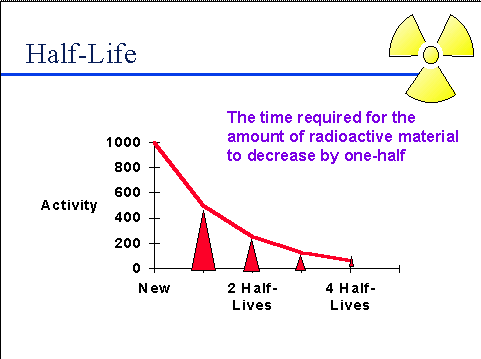
In this question (t½) is 45 minutes , which means that after 45 minutes half of the sample would have decayed and half would be left as it is. Let us start with 200g of the sample
After 45 minutes ( first half life) 200 /2 = 100 g decays and 100 g remains left.
After another 45 minutes ( two half lives or 90 minutes) 100 /2 = 50 g decays and 50 g remains left .
After another 45 minutes ( three half lives or 135 minutes or 2 hours and 15 minutes) 50 /2 = 25 g decays and 25 g remains left.
After three half lives or 135 minutes , 1/8 of the sample (25g) of the sample will be left.


