How many structural isomers of #C_3H_9N# exist?
1 Answer
Only 4. Watch the methyl groups move!
If you start with a three-carbon chain, that is 3 carbons and
Others involve moving methyl groups around. You can either move a methyl onto carbon 3 (forming the new carbon 2), or you can move it onto the nitrogen.
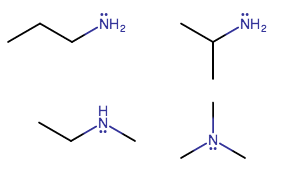
You can only move a methyl group onto one new carbon before you run into the nitrogen, giving one more structural isomer (isopropylamine).
Nitrogen can only accommodate two more methyl groups in place of its hydrogens before you run out of methyl groups to move, giving two more structural isomers (ethylmethylamine and trimethylamine). The former still has

