How many valence electrons does phosphorus have?
1 Answer
Nov 3, 2015
5
Explanation:
There are two ways to find out. Either you take a look at your periodic table and look at which group number
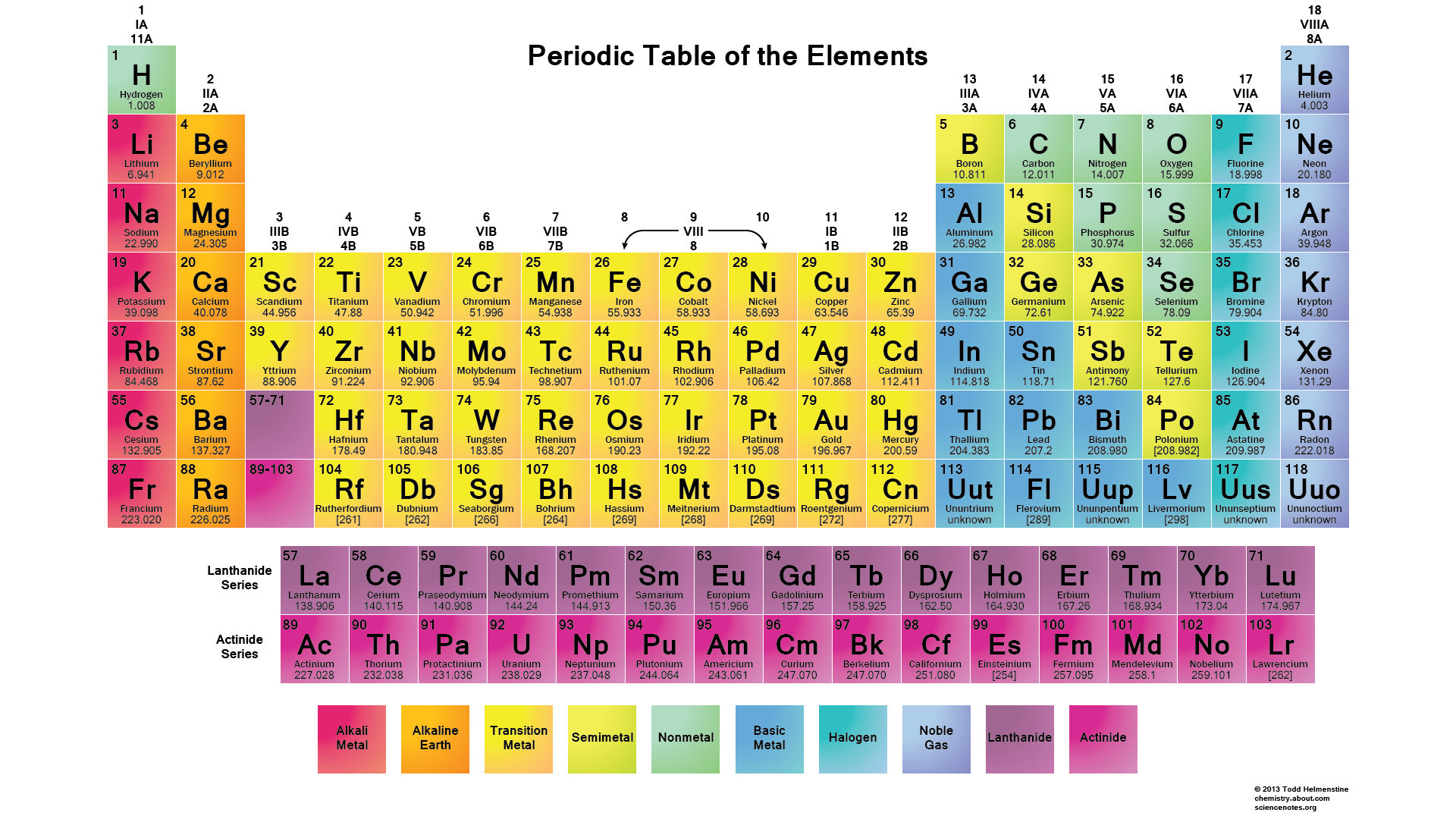
According to the periodic table above, phosphorus belongs to Group 5A. Therefore, Its valence electrons should be 5.
Let's check using the electron configuration:
The outermost orbitals,
Thus, valence electrons for

