How would you arrange the following elements in order of increasing ionization energy: Te, Pb, Cl, S, Sn?
2 Answers
Pb, Sn, Te, S, Cl
Explanation:
The ionization energy increases across a period but decreases down a group.
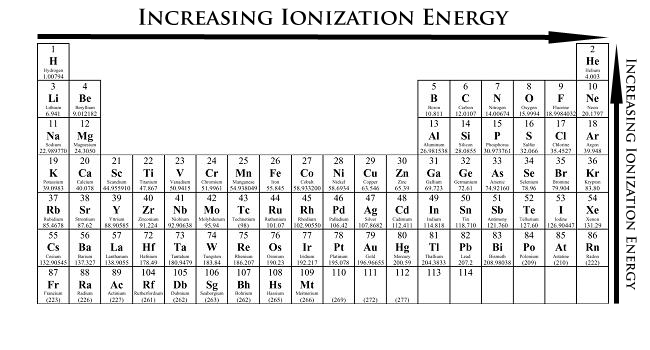
This means that the elements with the lowest ionization energies would be in the bottom left-hand corner of the periodic table. The change in ionization energies is also bigger going down the periodic table (by change within a group) than going across the periodic table (by change within a period).
So let's start from the bottom of the periodic table:
The period above (5) has two of the elements: Sn and Te. Well, since ionization energy increases across a period, Sn will have a smaller ionization energy than Te.
Now, let's go to the third period, where
The order is
Explanation:
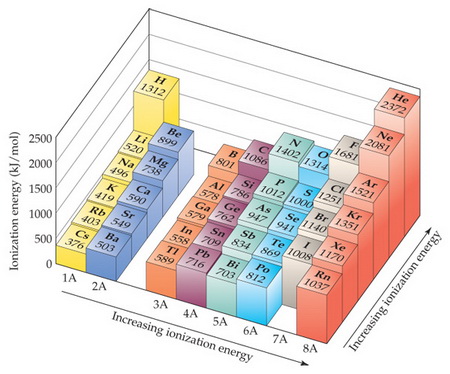
You have learned that ionization energy increases from top to bottom and from left to right in the Periodic Table.
You probably saw a diagram something like this.

Here's the portion of the Periodic Table that includes the elements in this question.

(Adapted from ZON PENA)
You would naturally predict the order to be
This is almost correct, but the correct order is
(From facebook)
Why is this so?
The electron configuration of
The electron configuration of
The
Thus the outer electrons experience a greater effective nuclear charge, and it is more difficult to remove them.
Hence
I hope that your instructor told you about this phenomenon before asking you to make a prediction.


