If potassium-40 undergoes radioactive decay by losing an electron, the daughter isotope is what?
1 Answer
Explanation:
First, make sure that you understand what happens when a radioactive nuclide decays by losing an electron - this is known as beta decay.
As you know, an electron, which is also known as a
The nucleus of an atom contains protons, which are positively charged particles, and neutrons, which are neutral particles.
When a
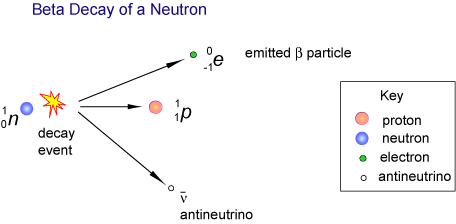
This means that the atomic number of the isotope, which as you know tells you how many protons are found in its nucleus, will increase by
On the other hand, the mass number of the isotope will remain unchanged, since a neutron is being converted to a proton.
This means that you can write
#""_19^40"K" -> ""_20^40"X" + beta^(-) + bar(nu)_e#
A quick look in the periodic table will show that element
Therefore, the daughter isotope that results from the beta decay of potassium-40 is calcium-40.
#""_19^40"K" -> ""_20^40"Ca" + beta^(-) + bar(nu)_e#

