If you decreased the volume of a sample of gas by a factor of three while maintaining a constant pressure, how would the absolute temperature of the gas be affected?
1 Answer
The temperature of the gas will decrease by a factor of
Explanation:
The pressure and temperature of a gas have a direct relationship when pressure and number of moles of gas are kept constant
Simply put, when the pressure of a given sample of gas is kept constant, increasing the volume of the gas will result in an increase in temperature
Similarly, decreasing the volume of the gas, as you have in your example, will result in a decrease in temperature.
Mind you, the temperature of the gas is actually its absolute temperature, i.e. its temperature expressed in Kelvin,
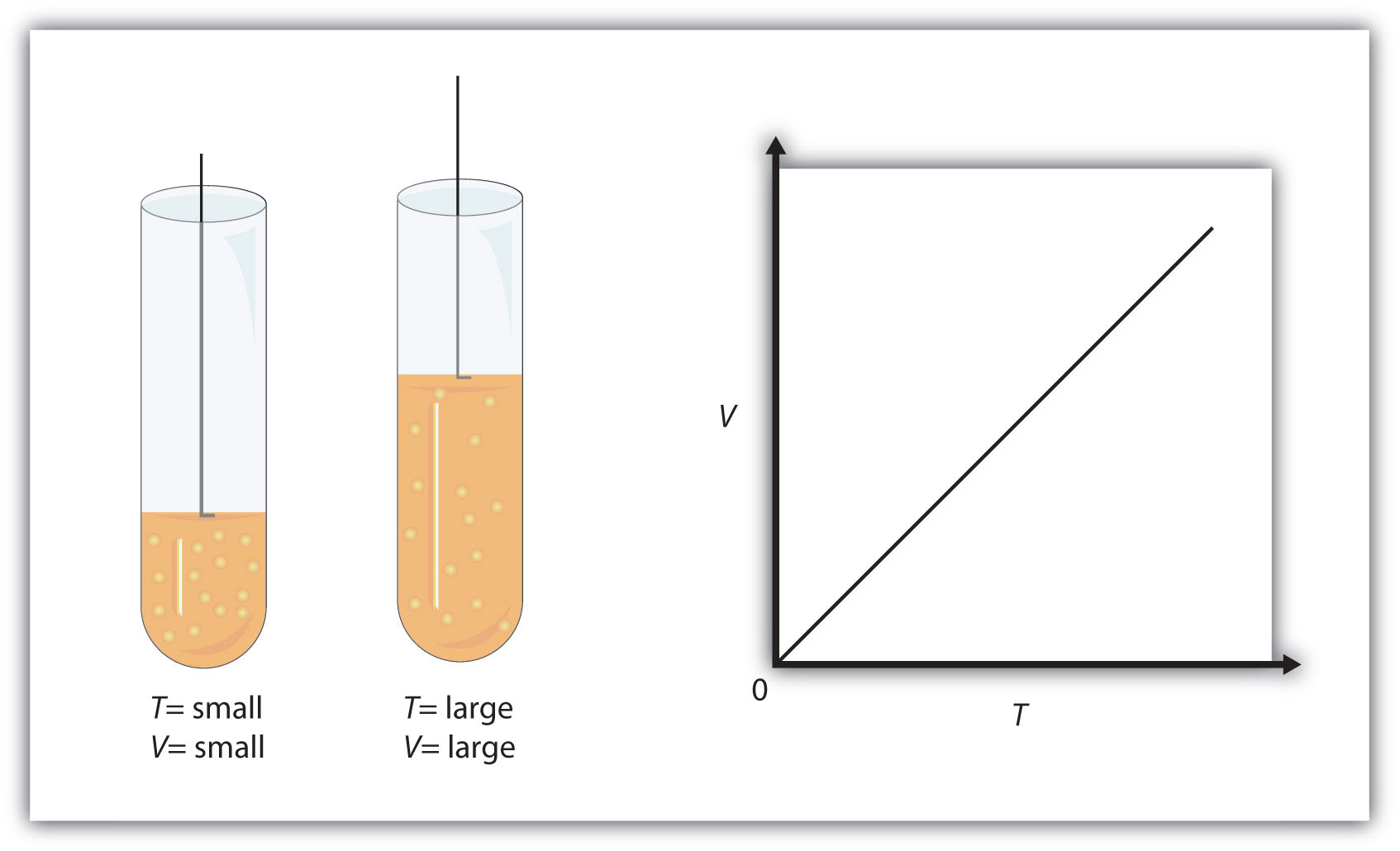
Now, the thing to remember here is that decreasing or increasing the volume of the gas by a factor
Mathematically, this can be shown by using the equation for Charles' Law
#color(blue)(|bar(ul(color(white)(a/a)V_1/T_1 = V_2/T_2color(white)(a/a)|)))" "# , where
Let's say that your starting volume is
#V_2 = 1/3 * V_1 -># the volume decreases by a factor of#3#
Rearrange the equation to solve for
#V_1/T_1 = V_2/T_2 implies T_2 = V_2/V_1 * T_1#
You will thus have
#T_2 = (1/3 * color(red)(cancel(color(black)(V_1))))/color(red)(cancel(color(black)(V_1))) * T_1 = color(green)(|bar(ul(color(white)(a/a)color(black)(1/3 * T_1)color(white)(a/a)|)))#
As you can see, decreasing the volume by a factor of

