In which state of matter do molecules have the highest kinetic energy?
2 Answers
As a gas, as it is here that the particles are moving fastest,
It would be a gas, because they lack the intermolecular forces that hold liquids together, and the rigidity that holds solids together.
This example of water pretty much summarizes it:
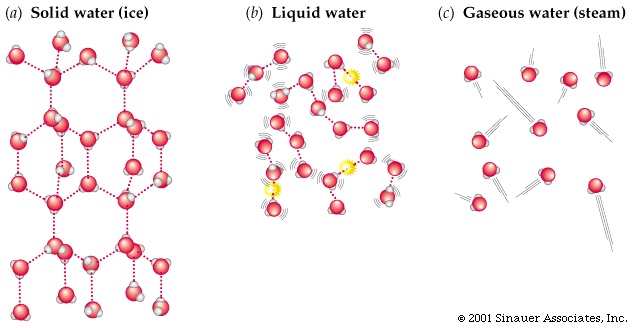
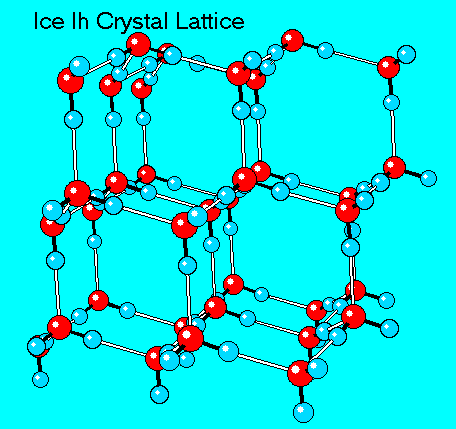
Solids, in general, are rigid. They are known to have a definite form, definite volume. Hence, their kinetic energy is smaller than that of loose fluids.
Ice is a convenient example:
A liquid, on the other hand, is generally more free-form - an indefinite form, but still a definite volume, as they aim to take the shape of their container.
Their intermolecular forces hold them together, such as the hydrogen-bonding networks in water, but that limits their motion less so than in comparable solids, as intermolecular forces aren't full bonds.
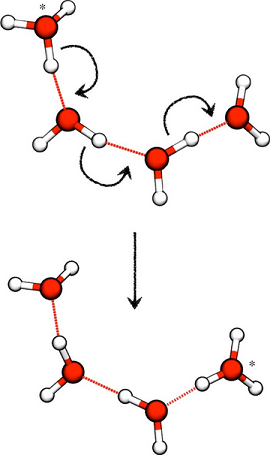
Although in water, let's say, proton-jumping throughout the water clusters is fair-game, the water molecules aren't as free to scatter into the air and disperse as in a gas. So, liquids have kinetic energies in between that of a comparable solid and gas.
Gases are free to move - they aren't necessarily lighter than air, per se, but they are expected to lack the intermolecular forces that hold liquids together, so they are more freely-moving.
Again:
- Solids are not freely-moving (definite volume), but not necessarily loose either (more definite shape than comparable liquids).
- Liquids are not freely-moving (that is, they have a definite volume), but they are fairly loose (an indefinite shape).
Hence, gases have more kinetic energy than comparable liquids, and those have more kinetic energy than comparable solids.


