Is ethoxide a better nucleophile in methanol or acetone?
1 Answer
Ethoxide is a better nucleophile in acetone.
Explanation:
The solvent affects the nucleophilicity of ethoxide ion.
A polar protic solvent like methanol can participate in hydrogen bonding with a nucleophile like ethoxide ion, creating a “shell” of solvent molecules around the ion.
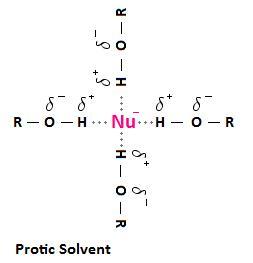
(from chemwiki.ucdavis.edu
This decreases the nucleophilicity of the ethoxide, because the nucleophile must push these solvent molecules out of the way in order to attack the substrate.
A polar aprotic solvent like acetone does not hydrogen bond to ethoxide ion to a significant extent.
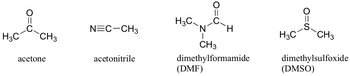
(from chemwiki.ucdavis.edu)
So the ethoxide ion is almost a "bare" nucleophile.
It is better able to attack the substrate and is therefore a stronger nucleophile.
WARNING: Ethoxide ion is also a strong base, so you will get a competing reaction in acetone — the aldol condensation.

