More reactive the metal, lesser the melting it has. Right?
1 Answer
It sounds like a coincidence to me. You're talking about chemical properties and trying to relate them to physical properties, which is usually not necessarily clear-cut.
Claim:
- Aluminum is more reactive than molybdenum, so it has a lower melting point?
Well, it IS more reactive, since it was more easily oxidized if the reaction is correct, and it DOES have a lower melting point... (
#1221^@ "F"# vs.#4753^@ "F"# )(Though you should consider that molybdenum (VI) oxide is a solid and not aqueous, and that the oxidation state of molybdenum in it is particularly large in comparison to aluminum ion.)
Counterexample:
- Aluminum is more reactive than zinc (more easily oxidized), so it SHOULD have a lower melting point than zinc, based on that logic. However:
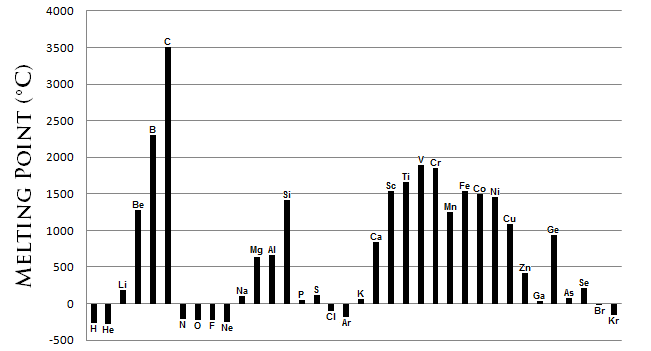
We can see that aluminum has a higher melting point than zinc.
(#1221^@ "F"# vs.#787.2^@ "F"# )
Therefore, reactivity and melting point are not related, or there are complicating factors that we're not considering. In other words, there is no clear link between reactivity and melting point.
Instead, I can think of a few physical reasons why aluminum has a lower melting point (a physical property).
Melting point for metals has to do with:
- How many electrons are "free" to move between metal atoms in the metallic crystal structure.
- How well they are delocalized throughout the metal atoms.
The more half-filled orbitals a metal has, the more electrons it can contribute
The following electron configurations for
#"Al": [Ne]3s^2 3p^1#
#"Mo": [Kr]5s^1 4d^5# (compare with#"Cr"# for the electron configuration, though#"W"# does not have the same valence electronic structure.)
Since
Furthermore, its significantly higher atomic number (
You can refer to this answer if you want to read further about melting points, or this page if you want to learn more about band theory.

