Normally, the pH of the human body is fixed in a very narrow range between 7.35 and 7.45. A patient with an acidotic blood pH of 7.3 may be treated with an alkali such as sodium bicarbonate. Why would this treatment raise the pH of the blood?
1 Answer
Here's what's going on here.
Explanation:
The main buffer system used to control the pH of the blood is the bicarbonate buffering system.
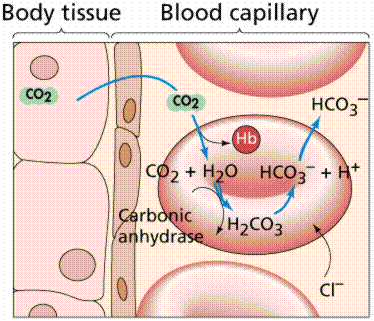
This buffer system revolves around the amount of carbon dioxide dissolved in the blood and on the amount of bicarbonate anions,
More specifically, it consists of carbonic acid,
#"H"_2"CO"_text(3(aq]) + "H"_2"O"_text((l]) rightleftharpoons "HCO"_text(3(aq])^(-) + "H"_3"O"_text((aq])^(+)#
Now, carbonic acid actually exists in equilibrium with dissolved carbon dioxide and water
#"CO"_text(2(aq]) + "H"_2"O"_text((l]) rightleftharpoons "H"_2"CO"_text(3(aq])#
This means that you can represent the bicarbonate buffering system as two equilibrium reactions
#"CO"_text(2(aq]) + "H"_2"O"_text((l]) rightleftharpoons "H"_2"CO"_text(3(aq]) + "H"_2"O"_text((l]) rightleftharpoons "HCO"_text(3(aq])^(-) + "H"_3"O"_text((aq])^(+)#
As you know, chemical equilibria are governed by Le Chatelier's Principle, which states that a system at equilibrium will respond to a disturbance in that equilibrium by shifting in such a way as to counteract that disturbance.
Now, the normal position for this equilibrium corresponds to a pH value that falls in the range
A lower pH value corresponds to a higher concentration of hydronium ions,
Sodium bicarbonate,
Treating the patient with sodium bicarbonate is equivalent to Increasing the concentration of bicarbonate anions in solution.
This means that the second part of the equilibrium will shift to the left, since that will consume both the added bicarbonate anions and the excess hydronium ions
#"H"_2"CO"_text(3(aq]) + "H"_2"O"_text((l]) rightleftharpoons "HCO"_text(3(aq])^(-) + "H"_3"O"_text((aq])^(+)#
A shift to the left is equivalent to saying that the reverse reaction will be favored, meaning that you will see an increase in the concentration of unionized carbonic acid.
Since the concentration of hydronium ions decreases as a result of this shift in the position of the equilibrium, the pH of the blood will increase.
According to the Henderson - Hasselbalch equation, the pH of a weak acid - conjugate base buffer is equal to
#color(blue)("pH" = pK_a + log( (["conjugate base"])/(["weak acid"])))" "# , where
This shows that a decrease in pH corresponds to a decrease in the ratio between the conjugate base and the weak acid.
Adding conjugate base increases this ratio, which will result in an increase in pH.

