Using the phase diagram for #CO_2#, what phase is carbon dioxide in at -60#"^@#C and 15 atm pressure?
1 Answer
Jul 20, 2017
Well, consult the phase diagram.
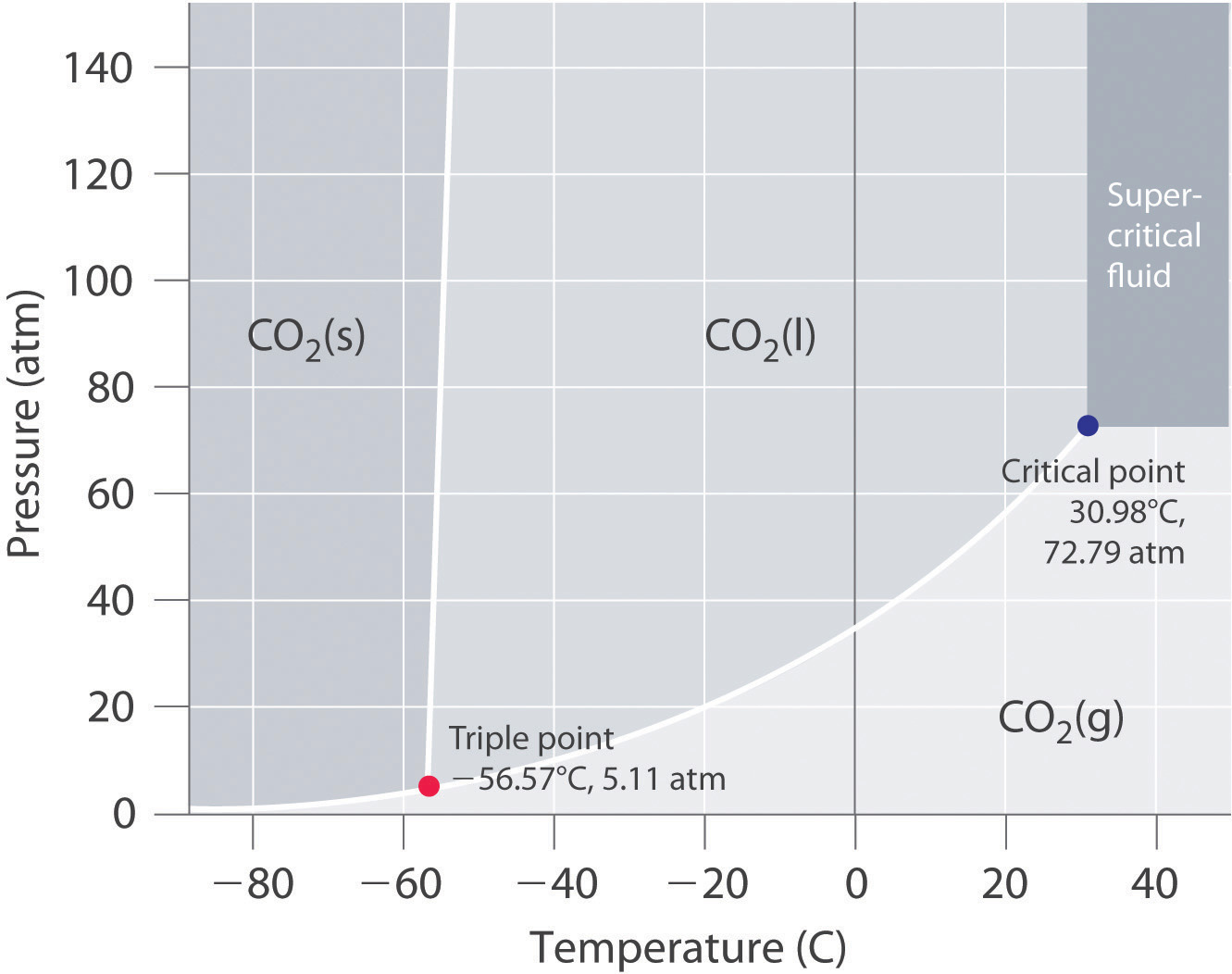
And then you literally just read it like any other graph: find your
#(x,y) -= (P,T) = ("15 atm", -60^@ "C")#
And one should easily see that
We call this dry ice; once obtained and brought into a laboratory, it spontaneously sublimes.

