What are dipole-dipole forces?
1 Answer
Dipole-dipole forces are the attraction between the positive end of one molecule and the negative end of another.
Explanation:
Dipoles form when there is a large difference in electronegativity between two atoms joined by a covalent bond.
The atoms share the electrons unequally because the more electronegative atom pulls the shared electrons toward itself.
That means that one atom has a partial negative charge, and the other atom has a partial negative charge.
For example, H-Cl has a dipole. The electrons spend more of their time near the Cl atom (the red end in the diagram below).

A dipole-dipole interaction is the attraction between two polar molecules. When they approach each other, the negative end one molecule attracts the positive end of the other.
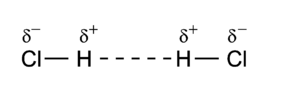
The molecules arrange themselves to increase their attraction and reduce their potential energy.


