What are intermolecular forces?
1 Answer
Intermolecular forces (IMFs) are attractive interactions between molecules. They lead to differences and sometimes trends in various physical properties.
These are typically listed in order of strength:
#"Dispersion" < "Dipole-Dipole" < "Hydrogen-bonding" < "Ion-Dipole" < "Ion Pairing"#
Hydrogen-bonding (which is not bonding) is shown below in an example diagram for
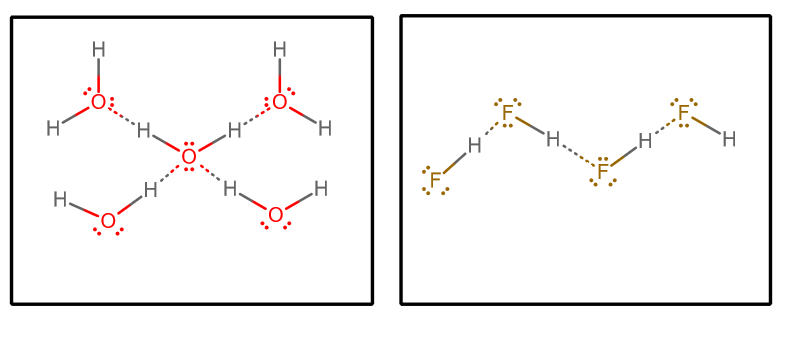
Other features which are equally important are described below.
IMFs span a spectrum of:
- temporary partial charge-partial charge interactions
- permanent partial charge-partial charge interactions
- permanent partial charge-full charge interactions
- permanent full charge-full charge interactions
Depending on electronegativities of each atom involved, the IMF strength and interaction time changes.
Here are tables organizing this into the known IMFs.
Table 1. Interaction Times
#ul("IMF"" "" "" "" "" "" "" ""Interaction Time")#
#" "" "" ""Dispersion"" "color(white)(....)"Temporary"#
#" "" ""Dipole-Dipole"" "" ""Permanent"#
#"Hydrogen-bonding"" "color(white)(....)"Permanent"#
#" "" "" ""Ion-Dipole"" "color(white)(....)"Permanent"#
#" "" "" ""Ion Pairing"" "" ""Permanent"#
Table 2. Degree of Charge Interactions
#ul("IMF"" "" "" "" "" "" "" ""Degree of Interaction")#
#" "" "" ""Dispersion"" "color(white)(....)"Partial/Partial"#
#" "" ""Dipole-Dipole"" "" ""Partial/Partial"#
#"Hydrogen-bonding"" "color(white)(....)"Partial/Partial"#
#" "" "" ""Ion-Dipole"" "color(white)(....)"Full/Partial"#
#" "" "" ""Ion Pairing"" "" ""Full/Full"#
Table 3. Typical Participants
#ul("IMF"" "" "" "" "" "" "" ""Participants"" "" "" "" "" "" ")#
#" "" "" ""Dispersion"" "" ""Nonpolar Molecules"#
#" "" ""Dipole-Dipole"" "color(white)(..)"Moderately Polar Molecules"#
#"Hydrogen-bonding"" "color(white)(..)"Molecules with XH bonds"^"*"#
#" "" "" ""Ion-Dipole"" "" ""Ions in Polar Solvent"#
#" "" "" ""Ion Pairing"" "color(white)(..)"Ions with large charges"#
#"*"# #-# #"X"# indicates a very electronegative atom, usually#"N"# ,#"O"# , or#"F"# . Note that these must be bonded DIRECTLY to#"H"# , not just be in the chemical formula. Note also that there are exceptions, like chloroform interacting with acetone, which do NOT seem to contain sufficiently electronegative atoms.

